|
"In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment. Answer = CF2Cl2 (Dichlorodifluoromethane) is Polar. Calculate the molecular polarity (polar, non-polar) of a chemical bond based. Calculate the electronegativity difference (ΔEN) and average ( EN) of the two electronegativities, and use the table below to determine the bond type and polarity. Nitrate – has a symmetrical …The bond polarity between two atoms can be estimated if you know the electronegativity of both elements. It consists of polar N-O and N=O bonds due to an electronegativity difference of 0.4 units between an oxygen and a nitrogen atom. View Available Hint (s) SiCl F SICH PCI SeF Nonpolar. Drag the appropriate items to their respective bins. View Available Hint (s) PB SF OF, NE Nonpolar Polar Determine whether each molecule is polar or nonpolar. Determine whether each molecule given below is polar or nonpolar.  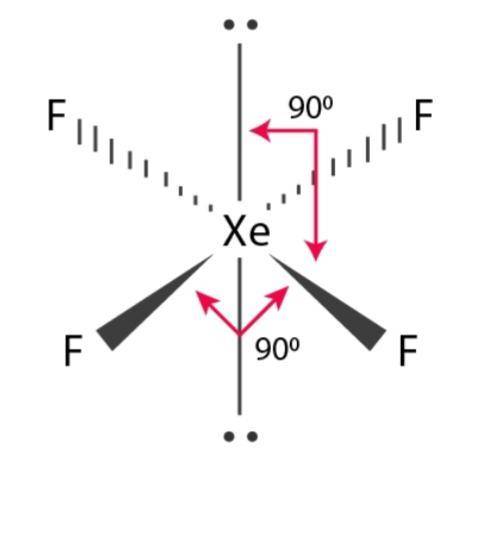
Nonpolar compounds will be symmetric, meaning all of the sides around the central atom are identical - bonded to the same element with no unshared pairs of electrons.Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom or having atoms. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. It is bonded to five atoms of fluorine (F) at the sides. Page Contents show How to draw lewis structure of IF5? The Lewis structure of iodine pentafluoride (IF5) consists of iodine (I) atom at the center.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
